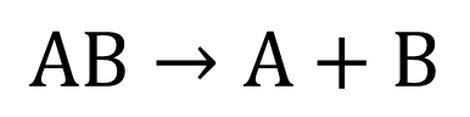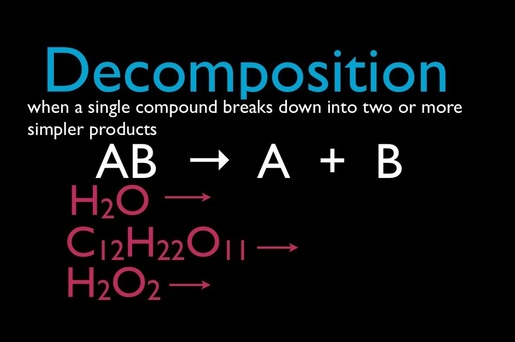The Merriam-Webster Dictionary defines decomposition as: "to cause something (such as a chemical) to be separated into smaller or simpler parts".
Decomposition reaction: A→B+C
A= Compound
B/C=Compound or element
For example, in the decomposition of sodium hydrogen carbonate (also known as sodium bicarbonate), sodium carbonate, carbon dioxide, and water are produced from the single substance sodium hydrogen carbonate.
2NaHCO3(s) → Na2CO3(s) + CO2(g) + H2O(ℓ)
Breaking It Down
Video
Decomposition reaction: A→B+C
A= Compound
B/C=Compound or element
For example, in the decomposition of sodium hydrogen carbonate (also known as sodium bicarbonate), sodium carbonate, carbon dioxide, and water are produced from the single substance sodium hydrogen carbonate.
2NaHCO3(s) → Na2CO3(s) + CO2(g) + H2O(ℓ)
Breaking It Down
Video